Fast shipping by DHL
Search suggestions
No search results.
Items
No search results.
Categories
No search results.
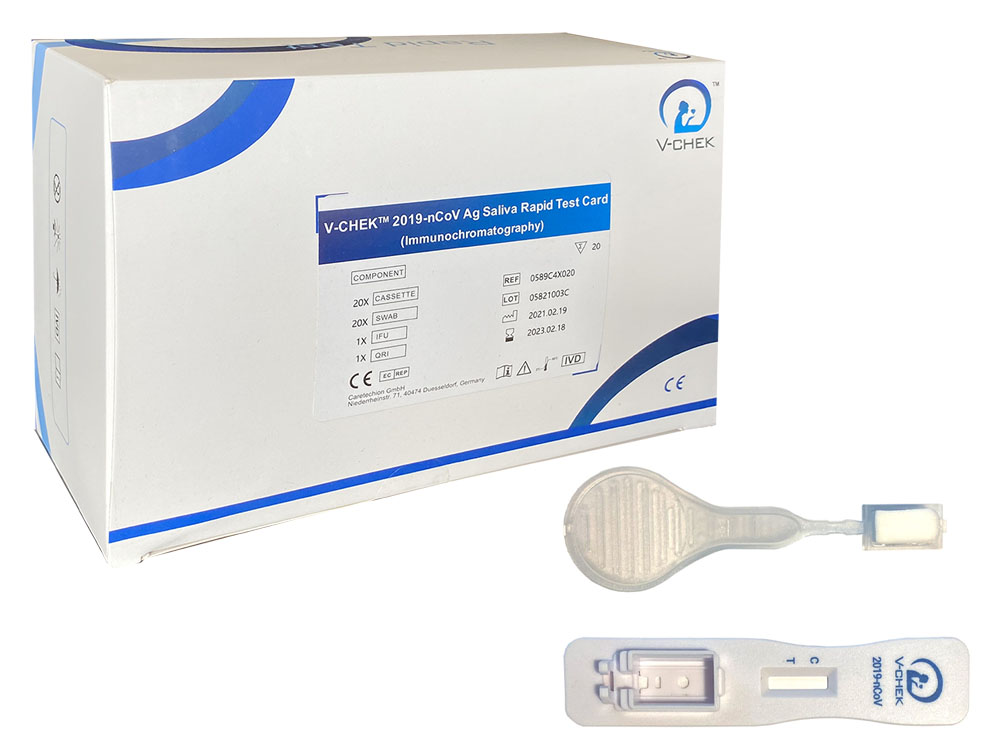
V-Check Antigen Corona Lollipop Rapid Test - 20 pcs.
- Item number: 262
- Manufacturer: V-Check

You have questions?
Our trained contact persons will be happy to advise and help you.
V-Check TM 2019-nCoV Saliva Antigen Corona Lollipop Rapid Test - 1 pack = 20 rapid tests.
Antigen saliva test for the qualitative detection of antigens within the first 7 days after symptom onset.
Details at a glance
20 saliva collectors included
bring test cassette to room temperature (18-26°C) before testing
For use by trained clinical laboratory personnel
The V-ChekTM 2019-nCoV Ag Saliva Lollipop Rapid Test (Immunochromatography) is a lateral flow immunoassay, for the qualitative in vitro detection of 2019-nCoV antigen. It is an immunochromatography sandwich assay for the qualitative determination of nucleocapsid protein antigen of 2019-nCoV in saliva samples. This assay is for medical laboratory use only or for immediate determination by medical personnel and is not for self-testing. It should not be used as the sole basis for diagnosis and is suitable for screening of the general population.
suitable
suitable for rapid testing of children and adolescents
easy to use as a "lollipop test
final result already after 10 minutes
BfArM test ID AT015/21
A positive test result must be further confirmed. A negative test result cannot exclude an infection.
The V-ChekTM 2019-nCoV Ag Saliva Rapid Test is based on a double antibody sandwich to detect the antigen of the novel coronavirus 2019-nCovid antigen in saliva samples. During the test, the gold-labeled anti-2019-nCoV monoclonal antibody in the label pad binds to the 2019-nCoV antigen in the sample, forming a complex. This reaction complex moves along the nitrocellulose membrane during the chromatography procedure and is detected by the anti-2019 nCoV monoclonal antibody with which the nitrocellulose membrane is pre-coated in the test pad (T), and finally a red color reaction line is formed in the T zone. If the sample does not contain 2019nCoV antigen, no red reaction line can form in the T zone. Regardless of whether the specimen to be tested contains the 2019-nCoV antigen, a red reaction line will always form in the control region (C).
Read the instructions for use carefully.
Preparation
Bring the test cassette to room temperature (18-26°C) before testing. Do not eat or consume food for two hours prior to testing.
Specimen Collection
Insert the sponge end of the saliva collector into the mouth. Gently wipe along the inside of the mouth and on the tongue to collect saliva (approximately 90 seconds). If possible, allow the saliva to soak into the sponge by itself. Remove the saliva collector from the mouth when the sponge is completely saturated and soft or the indicator on the back has turned blue. The specimen should be used as soon as possible after collection. Specimens should be tested immediately after collection. Samples should not be inactivated. The results of the V-Check TM 2019-nCoV Antigen Saliva Test should not be considered a confirmed diagnosis; they are for clinical reference only. Assessment should be made based on the combination of RT-PCR test results, clinical symptoms, epidemiologic information, and other clinical data.
Sensitivity, specificity and accuracy
The performance of the V-Check TM 2019-nCoV Antigen Saliva Test was demonstrated using 243 samples from symptomatic patients tested within 7 days of symptom onset. RT-PCR is used as the reference method for Covid-19 antigen saliva testing. Specimens were considered positive if RT-PCR yielded a positive result. Samples were considered negative if RT-PCR produced a negative result.
Relative sensitivity 95.65% (95%CI*: 90.22%-98.13%)
Relative specificity 98.44% (95%CI*: 94.48%-99.57%)
Accuracy 97.12% (95%CI*: 94.17%-98.60%)
*confidence interval
Storage and stability
The V-Check TM 2019-nCoV Antigen Saliva Test may be stored at room temperature or refrigerated (2-30°C). Until use, the test cassette must remain in the sealed pouch. Use the test cassette and saliva collector only until the expiration date printed on the package. Do not freeze. Do not use after the expiration date.
Materials included
20 test cassettes
20 saliva collectors/sponges
1 instruction manual
1 brief description
Additionally required: Timer (not included)
For further information, please refer to the instructions for use.
For professional use only.
- Item number: 262
- Weight: 367g
- Content: 20 piece
- Dimensions: 210x90x140mm
- Manufacturer: V-Check
- Condition: New
Other customers also bought
 Add to wish list
Detects Omicron
(0)
Ultimed
ULTIMED Lollipop Covid-19 Rapid Test €46.13
Add to wish list
Detects Omicron
(0)
Ultimed
ULTIMED Lollipop Covid-19 Rapid Test €46.13 €50.34-8% €46.13€50.34-8% €2.31 / piece €2.31 / piece Delivery time on request Add to wish list Add to wish list
Layman-Selftest
Detects Omicron
(1)
AmonMed
AmonMed lollipop selftest - CE1434 €1.59
Add to wish list
Layman-Selftest
Detects Omicron
(1)
AmonMed
AmonMed lollipop selftest - CE1434 €1.59 €1.67-5% €1.59€1.67-5% Delivery time on request Add to wish list Add to wish list
Layman-Selftest
Detects Omicron
(1)
Wantai
WANTAI Nasal Swab & Saliva (Lollipop) Selftest - CE2854 €0.83
Add to wish list
Layman-Selftest
Detects Omicron
(1)
Wantai
WANTAI Nasal Swab & Saliva (Lollipop) Selftest - CE2854 €0.83 €1.25-33% €0.83€1.25-33% Delivery time on request Add to wish list Add to wish list
Detects Omicron
(0)
Safecare
SAFECARE® Corona Lolli Antigen Rapid Test - 25 pcs. €23.11
Add to wish list
Detects Omicron
(0)
Safecare
SAFECARE® Corona Lolli Antigen Rapid Test - 25 pcs. €23.11 €29.33-21% €23.11€29.33-21% €0.92 / piece €0.92 / piece Delivery time on request Add to wish list
This might also interest you
Shipping costs
- Germany 5.80€
- Europe from 15,99€
- Switzerland from 26,99€
DHL Parcel
| bis 5kg | bis 10kg | bis 20kg | bis 31,5kg | |
| 15,99€ | 20,99€ | 31,18€ | 43,86€ | |
| 26,90€ | 34,99€ | 48,99€ | 55,99€ | |
| 29,99€ | 37,99€ | 52,99€ | 60,99€ |
DHL Express Parcel
| bis 5kg | bis 10kg | bis 20kg | bis 31,5kg | |
| 19,19€ | 25,19€ | 37,39€ | 52,59€ | |
| 34,90€ | 44,90€ | 59,90€ | 69,90€ | |
| 39,90€ | 49,90€ | 54,90€ | 74,90€ | |
| 39,90€ | 49,90€ | 69,90€ | 79,90€ | |
| 49,90€ | 69,90€ | 99,90€ | 129,90€ | |
| 49,90€ | 54,90€ | 89,90€ | 124,90€ | |
| 54,90€ | 74,90€ | 119,90€ | 159,90€ | |
| 64,90€ | 84,90€ | 129,90€ | 169,90€ |
all prices incl. VAT