Fast shipping by DHL
Search suggestions
No search results.
Items
No search results.
Categories
No search results.
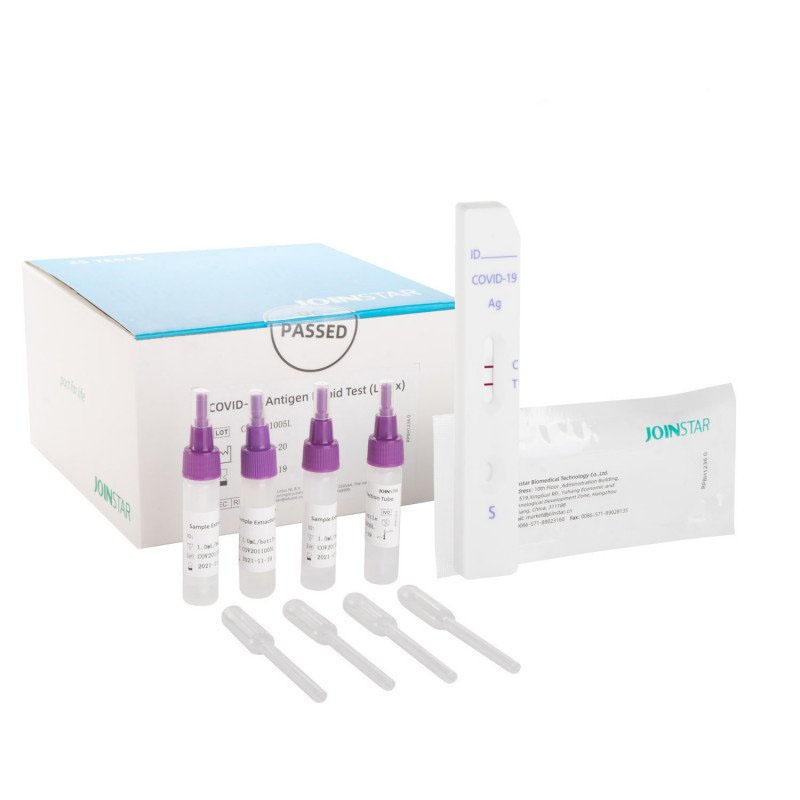
JOINSTAR® COVID-19 Antigen Rapid Test Saliva, Spit Test - 25 pcs.
- Item number: 235
- Manufacturer: Joinstar

You have questions?
Our trained contact persons will be happy to advise and help you.
Spit Test Antigen Rapid Test Kit - Saliva (Oropharyngeal Saliva / Sputum) SARS-CoV-2
(Non-invasive sampling - easy to use with saliva)
Home tests, tests for institutions (e.g. companies and schools) for carefree resumption of professional and educational activities.
World's most innovative antigen test
Very accurate due to high affinity between ACE2 receptors and S-proteins
Sensitive to different mutants
No cross reactivity
NON-INVASIVE PROBATE (saliva)
No medical supervision required
For immediate detection of infection
Result in approx. 10-15 minutes
All necessary materials included
BfArM-listed and reimbursable
Listed by the German Federal Institute for Drugs and Medical Devices in the "List of Antigen Tests for Direct Pathogen Detection of Coronavirus SARS-CoV-2".
Manufacturer: JOINSTAR Biomedical Technology Co., Ltd
Sensitivity and Specificity
The COVID-19 antigen rapid test was tested with the leading commercial reagent (PCR) and the results showed that the COVID-19 test has high sensitivity and specificity.
The following 3 sampling options are available:
Saliva sample of the posterior oropharynx (mouth part of the pharynx).
Relative sensitivity: 90.00% (95%CI: 79.49% - 98.96%)
Relative specificity: 100.00% (95%CI: 88.43% - 100.00%)
Accuracy: 96.67% (95%CI: 86.05% - 97.51%)
Sputum sample (secretion of the deep airways that enters the throat during coughing)
Relative sensitivity: 95.00% (95%CI: 86.08% - 96.24%)
Relative specificity: 100.00% (95%CI: 88.43% - 100.00%)
Accuracy: 96.67% (95%CI: 90.57% - 99.31%)
Stool sample
Relative sensitivity: 95.00% (95%CI: 86.08% - 96.24%)
Relative specificity: 100.00% (95%CI: 88.43% - 100.00%)
Accuracy: 96.67% (95%CI: 90.57% - 99.31%)
MATERIAL PROVIDED
per box of 25
25x test cassettes
25x sampling tubes
25x pipette
25x disposable cups
1x instruction manual (enclosed text)
Not included in delivery: stool sample collector (if needed)
Test Principle:
The novel coronavirus invades human cells through the specific binding of its spike glycoprotein (binding protein) to the ACE2- receptor located on the human cell membrane. In this assay, the ACE2 receptor was replaced by antibody to develop a novel binding protein receptor chromatography assay for the rapid detection of the novel coronavirus. In clinical practice, the test can be used for rapid detection of SARS- CoV-2 and all its mutations in saliva samples from the posterior oropharyngeal region, sputum and stool samples. The test takes only 15 minutes to perform and is much simpler than that of an RT-PCR test. The SARS-CoV-2 virus has been found to have evolved even more contagious mutations due to mutations in S1 proteins (such as D614G) that have stronger binding to ACE2 receptors. Given the principle of the test, which is based on the binding of ACE-2 receptors, the test should be able to detect such mutations as well.
The test kit contains a nitrocellulose membrane (NC) that has a coating of rabbit anti-S1- proteins of coronavirus in the test line region (T) and a coating of polyclonal goat anti-rabbit IgG- antibodies in the control region (C). The latex-labelled ACE2 protein and the latex-labelled rabbit IgG are embedded in the reagent pad.
To perform the assay, three drops of sample are added to the sample well and the sample flows upward by capillary action. After a 15-minute incubation period, the latex-labeled ACE2 protein is bound by the S1 protein of the virus and then captured by anti-S1 protein antibodies on the test line region if the sample contains the virus. If the sample does not contain the virus, then the latex-labeled ACE2 protein is not captured by the anti-S1 protein antibodies coated on the T-line region, so no T-line appears. Whether or not the sample contains virus, the latex-labeled rabbit IgG will react with the polyclonal goat anti-rabbit IgG antibodies on the control region (C) and a colored line will appear in the control region.
Once the assay is complete, the amount of latex ACE2 protein bound to the T-line is directly related to the concentration of novel coronavirus in the sample, whereas the amount of latex bound to the control region is not related to the concentration of coronavirus in the sample.
STORAGE AND STABILITY
The test is stable for 12 months if all components remain in the sealed pouch and the test is stored away from light at 2° Celsius.
- Item number: 235
- Weight: 1g
- Content: 25 piece
- Manufacturer: Joinstar
- Condition: New
Other customers also bought
 Add to wish list (0)
Realy-Tech
Realy Tech Spit Test Antigen SARS-COV-2 - (20 pcs) €54.54 €54.54 €2.73 / piece €2.73 / piece Delivery time on request Add to wish list
Add to wish list (0)
Realy-Tech
Realy Tech Spit Test Antigen SARS-COV-2 - (20 pcs) €54.54 €54.54 €2.73 / piece €2.73 / piece Delivery time on request Add to wish list Add to wish list
Detects Omicron
(0)
Anbio-Biotech
Anbio Biotech Antigen Rapid Test 3in1 - 20 pcs. €15.03
Add to wish list
Detects Omicron
(0)
Anbio-Biotech
Anbio Biotech Antigen Rapid Test 3in1 - 20 pcs. €15.03 €16.72-10% €15.03€16.72-10% €0.75 / piece €0.75 / piece Delivery time on request Add to wish list Add to wish list
Detects Omicron
(3)
Clungene
CLUNGENE® COVID-19 antigen saliva test - 20 pcs. €30.24
Add to wish list
Detects Omicron
(3)
Clungene
CLUNGENE® COVID-19 antigen saliva test - 20 pcs. €30.24 €33.53-9% €30.24€33.53-9% €1.51 / piece €1.51 / piece Delivery time on request Add to wish list
This might also interest you
Shipping costs
- Germany 5.80€
- Europe from 15,99€
- Switzerland from 26,99€
DHL Parcel
| bis 5kg | bis 10kg | bis 20kg | bis 31,5kg | |
| 15,99€ | 20,99€ | 31,18€ | 43,86€ | |
| 26,90€ | 34,99€ | 48,99€ | 55,99€ | |
| 29,99€ | 37,99€ | 52,99€ | 60,99€ |
DHL Express Parcel
| bis 5kg | bis 10kg | bis 20kg | bis 31,5kg | |
| 19,19€ | 25,19€ | 37,39€ | 52,59€ | |
| 34,90€ | 44,90€ | 59,90€ | 69,90€ | |
| 39,90€ | 49,90€ | 54,90€ | 74,90€ | |
| 39,90€ | 49,90€ | 69,90€ | 79,90€ | |
| 49,90€ | 69,90€ | 99,90€ | 129,90€ | |
| 49,90€ | 54,90€ | 89,90€ | 124,90€ | |
| 54,90€ | 74,90€ | 119,90€ | 159,90€ | |
| 64,90€ | 84,90€ | 129,90€ | 169,90€ |
all prices incl. VAT